  (206).jpg)
In this model n = ∞ corresponds to the level where the energy holding the electron and the nucleus together is zero. why does the Rydberg equation work for hydrogen but not for helium unlike hydrogen, helium has more than one electron. Study with Quizlet and memorize flashcards containing terms like Which of these questions were left unanswered by Rutherfords atomic model, Light emitted by elements when heated revealed the arrangement of in atoms which explained the behavior of elements. Where all wavelengths and frequencies are represented (rainbow) The emitted light corresponds to. 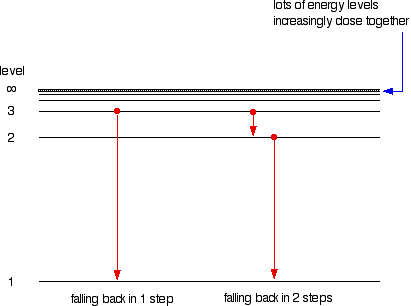
The photon energy of the emitted photon is equal to the energy difference between the two states. a single wavelength of light that can be seen when the light from an excited element is passed through a prism Spectrum. \) is the Rydberg constant in terms of energy, Z is the atom is the atomic number, and n is a positive integer corresponding to the number assigned to the orbit, with n = 1 corresponding to the orbit closest to the nucleus. The emission spectrum of a chemical element or chemical compound is the spectrum of frequencies of electromagnetic radiation emitted due to an electron making a transition from a high energy state to a lower energy state. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
